INTRODUCTION
Lymphangioleiomyomatosis (LAM) is a rare disease characterized by the proliferation of the atypical smooth muscle cells of the bronchioles, alveolar septa, perivascular spaces, and lymphatics, resulting in obstruction of the airways, which leads to pulmonary cyst formation [1]. The two most common symptoms of LAM are pneumothorax and dyspnea on exertion [2]. In this study, we report a case of a 41-year-old woman who presented with recurrent pneumothorax caused by an unexpected LAM.
CASE REPORT
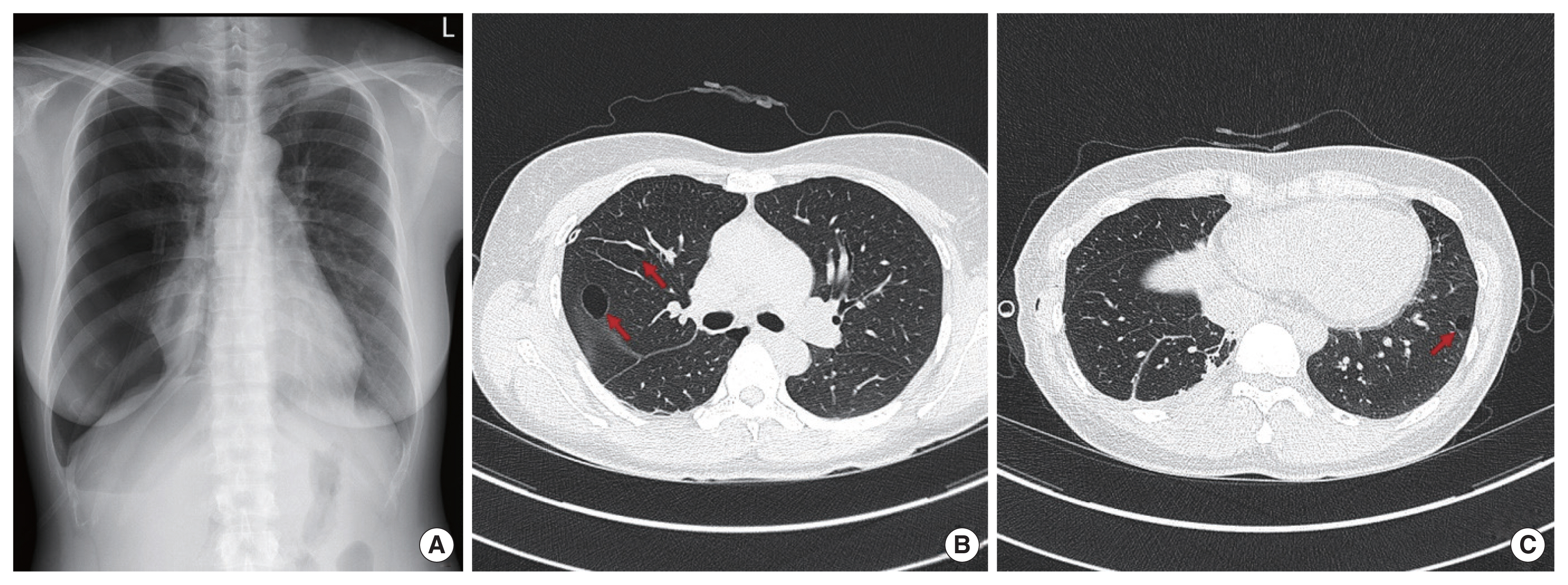
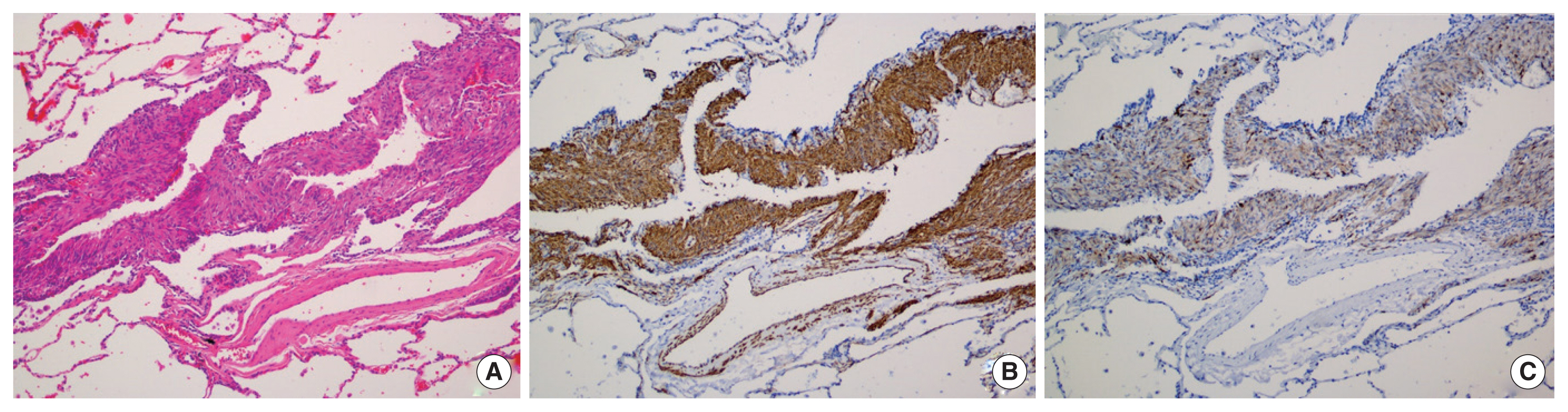
A 41-year-old woman, who presented with acute pain in her right chest, was diagnosed with recurrent pneumothorax based on her chest radiography (Fig. 1A). The patient had undergone a thoracostomy for pneumothorax using a 7F catheter 1 year before presentation. All episodes of pneumothorax occurred during her menstruation. She denied any history of dyspnea on exertion and usually lived a healthy lifestyle. The patient had never smoked. We performed a thoracostomy using a 7F catheter to relieve her symptoms. Chest computed tomography (CT) performed after thoracostomy showed a few round cystic lesions with thin walls in both lungs (Fig. 1B, C). We suspected that her pneumothorax was catamenial pneumothorax rather than LAM because the chest CT of patients with LAM we experienced in the past showed multiple thin-walled cysts homogeneously distributed in both lungs. The patient underwent video-assisted thoracic surgery (VATS) which showed that her lungs were entirely normal in appearance, except for the presence of bullae at the base of the right upper lobe, which was resected using a thoracoscopic stapler (Fig. 2). Histological examination of the surgical specimen revealed that the bullae could be attributed to LAM. The specimen showed proliferative spindle cells in the cyst wall, which were immunohistochemically positive for alpha-smooth muscle actin and anti-human melanin black 45 antibodies (Fig. 3). The patient was discharged 2 days after surgical management, with an uneventful recovery, and was referred to a specialized facility for appropriate management 2 weeks postoperation. The patient provided written informed consent for the publication of clinical details and images.
DISCUSSION
LAM is a rare condition in which the proliferation of atypical smooth muscle cells leads to progressive cystic destruction of the lungs. It predominantly affects women of childbearing age [1]. Chest CT is often diagnostic, showing multiple cysts of similar shape, size, and homogeneous distribution throughout the lungs. Cysts are typically thin-walled, rounded, or oval-shaped and measure between 2 mm and 10 mm, most of them are <30 mm [3]. CT grading measures the extent of lung involvement with LAM. Grade 1 is a mild disease with less than one-third of the lungs having cysts. Grade 2 is a moderate disease with one-to two-thirds of the lungs having cysts. Cyst involvement in more than two-thirds of the lungs is a severe condition [4]. In our case, we found only a few cysts with well-preserved lung parenchyma. Before histological examination revealed the diagnosis, we did not suspect the possibility of LAM because of the presence of very few cysts. Avila et al. created a global category of minimal disease (grade 0), which includes patients with very few cysts [4]. Steagall et al. [5] found that cyst size was correlated with pneumothorax, and larger cysts were associated with greater numbers of pneumothorax. In our case, although the extent of lung involvement with LAM was minimal (grade 0), the size of resected cyst was more than 20 mm on CT.
In patients with LAM, the pulmonary function test (PFT) may be significant if there is a decrease in the forced expiratory volume in 1 second (FEV1) and diffusion capacity for carbon monoxide [1]. Patients with a history of pneumothorax have worse pulmonary function than patients without a history of pneumothorax [6]. Steagall et al. [5] reported that the rate of decline in FEV1 was higher in patients with pneumothorax in the early stage of LAM than in those without pneumothorax in the early stage of LAM. In our case, the patient did not undergo preoperative PFT because PFT was not routinely performed in previously asymptomatic patients who were planned to undergo wedge resection in our hospital. In addition, the lung parenchyma on chest CT was normal in appearance, except for the presence of a few cysts.
Repeated pneumothorax may cause acute exacerbation of LAM [6]. Due to the high recurrence rate of pneumothorax in patients with LAM, pleurodesis is suggested for patients after the first episode of pneumothorax [1]. A recent study showed that sirolimus, a mammalian target of rapamycin inhibitor, significantly reduced the risk of pneumothorax recurrence in patients with LAM [6]. In our case, we only performed pulmonary wedge resection without pleurodesis because we did not consider the possibility of LAM. We did not know whether she was receiving sirolimus because the patient was referred to a tertiary hospital. Although sirolimus is effective in stabilizing lung function and reducing the recurrence of pneumothorax, it is not curative [6,7]. Lung transplantation is considered the only hope for a cure in patients with severe LAM [1].
In conclusion, early diagnosis and prevention of repeated pneumothorax play important roles in avoiding lung transplantation. Knowledge of LAM helps thoracic surgeons diagnose LAM early, even if only a few cysts are found on chest CT in patients with pneumothorax.