A Rare Radiological Presentation of Pulmonary Mucosa-Associated Lymphoid Tissue Lymphoma as Bronchovascular Thickening and Ground Glass Opacities with Concurrent Pancreas Involvement
Article information
Abstract
Pulmonary mucosa-associated lymphoid tissue (MALT) lymphoma is a rare disease and usually presents as pulmonary masses, mass-like consolidation, or pulmonary nodules on chest images. We report a case of a 43-year-old man with symptoms of chronic cough for 1 year, showing bilateral diffuse bronchovascular bundle thickening and focal ground glass opacities on a chest computed tomography scan. Video-assisted thoracoscopic surgery was performed and the final pathologic diagnosis was pulmonary MALT lymphoma. Concurrent involvement of the pancreas was discovered during staging workup. After diagnosis, he was treated with cytotoxic chemotherapy and rituximab and showed improvements in his lung lesion and pancreas.
INTRODUCTION
Primary pulmonary lymphomas are a very rare cause of lung tumours (0.5%–1% of primary pulmonary malignancies). The most common primary pulmonary lymphoma is mucosa-associated lymphoid tissue (MALT) lymphoma (70%–90%) [1].
The most common chest computed tomography (CT) finding of pulmonary MALT lymphoma is multiple bilateral lung nodules with air bronchogram (90%), while ground glass opacities (GGO) and interlobular septal thickening are rare [2]. Especially rare findings of bronchovascular bundle thickening and GGO of pulmonary MALT lymphoma on chest CT scans can be found in pulmonary sarcoidosis, pulmonary interstitial edema, certain types of pneumonias like mycoplasma pneumonia, and usually show on diffuse interstitial lung disease. Thus, the risk of misdiagnosis of pulmonary MALT lymphoma can occur. Here, we report a case of pulmonary MALT lymphoma presenting with diffuse bilateral bronchovascular thickening and focal GGO with concurrent rare pancreas involvement, which was diagnosed by biopsy with video-assisted thoracoscopic wedge resection and successfully treated with cytotoxic chemotherapy and rituximab.
CASE REPORT
A previously healthy nonsmoker 43-year-old man visited Soonchunhyang University Cheonan Hospital with complaints of chronic dry cough for 1 year. He had no systemic symptoms such as fever, night sweats, or weight loss. A CT scan of the chest showed diffuse bilateral bronchovascular bundle thickening with focal GGO in the right middle lobe lateral segment and left lower lobe medial basal segment with interlobular septal thickening (Fig. 1).
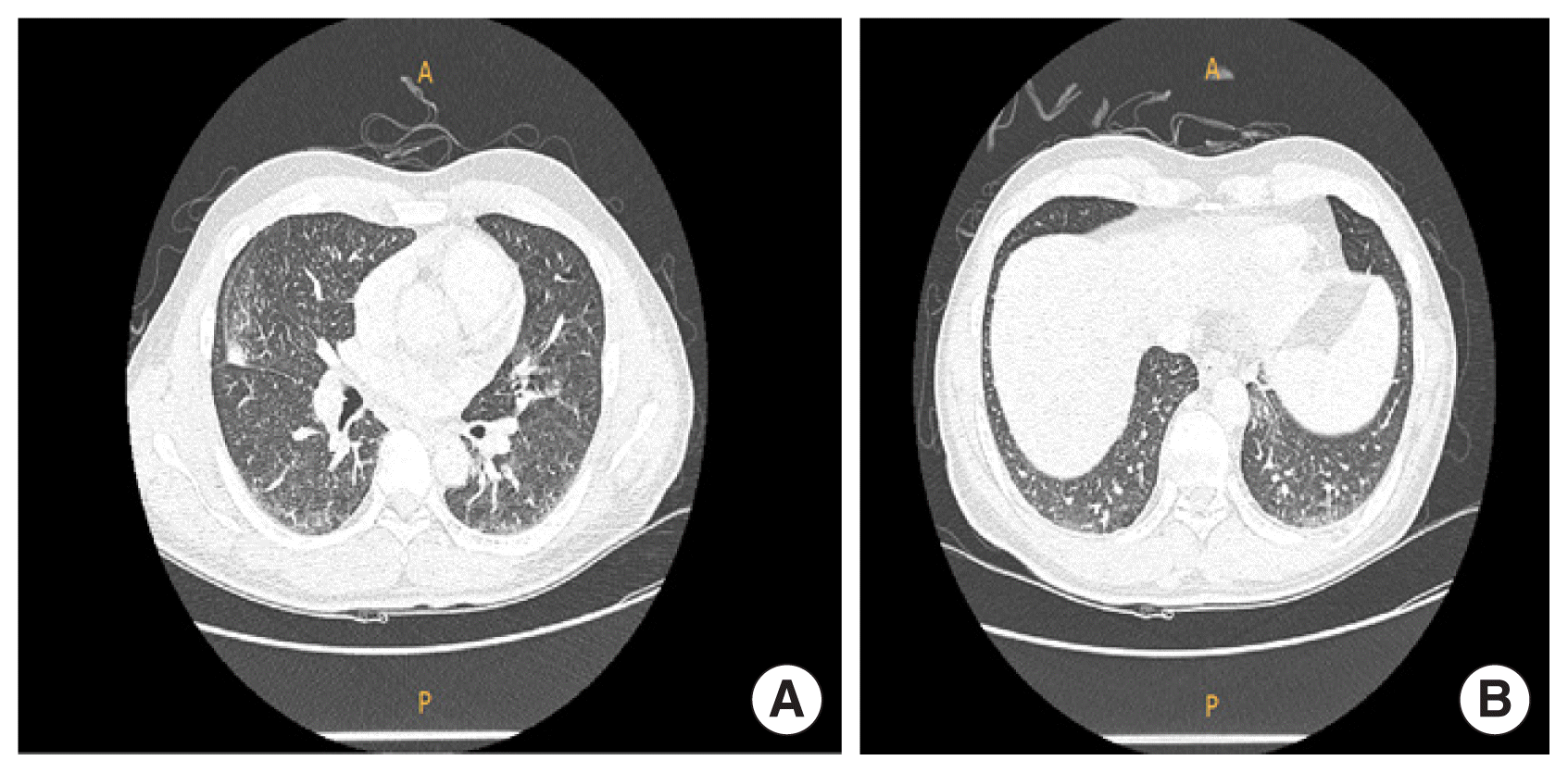
(A) Chest computed tomography showed bronchovascular bundle thickening with focal ground glass opacities in right middle lobe and (B) medial base of left lower lobe with interstitial septal thickening. A, anterior; P, posterior.
Laboratory data were within normal limits except mild elevation of eosinophil count and C-reactive protein, which were 0.79 × 103/μL and 6.08 mg/L (normal range, 0–5 mg/L), respectively. Human immunodeficiency virus (HIV) antibody results were negative. Because chest images included GGO and bronchovascular bundle thickening, we considered pulmonary sarcoidosis initially. During bronchoscopy, there was no endobronchial lesion and bronchoalveolar lavage fluid cell analysis showed a composition of 7% lymphocytes, 40.5% neutrophils, and 2.5% eosinophils and macrophages. Microbiologic tests for respiratory viruses, bacteria, and Mycobacterium tuberculosis were negative. Autoantibody tests for connective tissue disease-associated interstitial lung disease were also negative.
Video-assisted thoracoscopic wedge resection for lung biopsy was done at the right middle lobe. A scan view of resected lung specimen exhibited multiple nodular infiltration of dense inflammatory cells around bronchovascular structures (Fig. 2). On high power examination, lymphocytes invaded into vascular structures and bronchial epithelium, so called lymphoepithelial lesions. Immunohistochemistry for CD20, a marker for B cells, was positive for infiltrating lymphocytes. According to pathological findings, the patient was finally diagnosed with pulmonary MALT lymphoma.

(A) A scan view of resected lung specimen exhibited multiple nodular infiltration of dense inflammatory cells around bronchovascular structures (H&E, × 4). (B) On high power examination, lymphocyte infiltration (arrows) was noted in bronchus, forming the lymphoepithelial lesions (H&E, × 400). (C) Immunohistochemistry for pancytokeratin highlighted the lymphoepithelial lesions (pancytokeratin, × 400). (D) Infiltrating lymphocytes showed immunoreactivity for CD20 (arrows) (CD20, × 400).
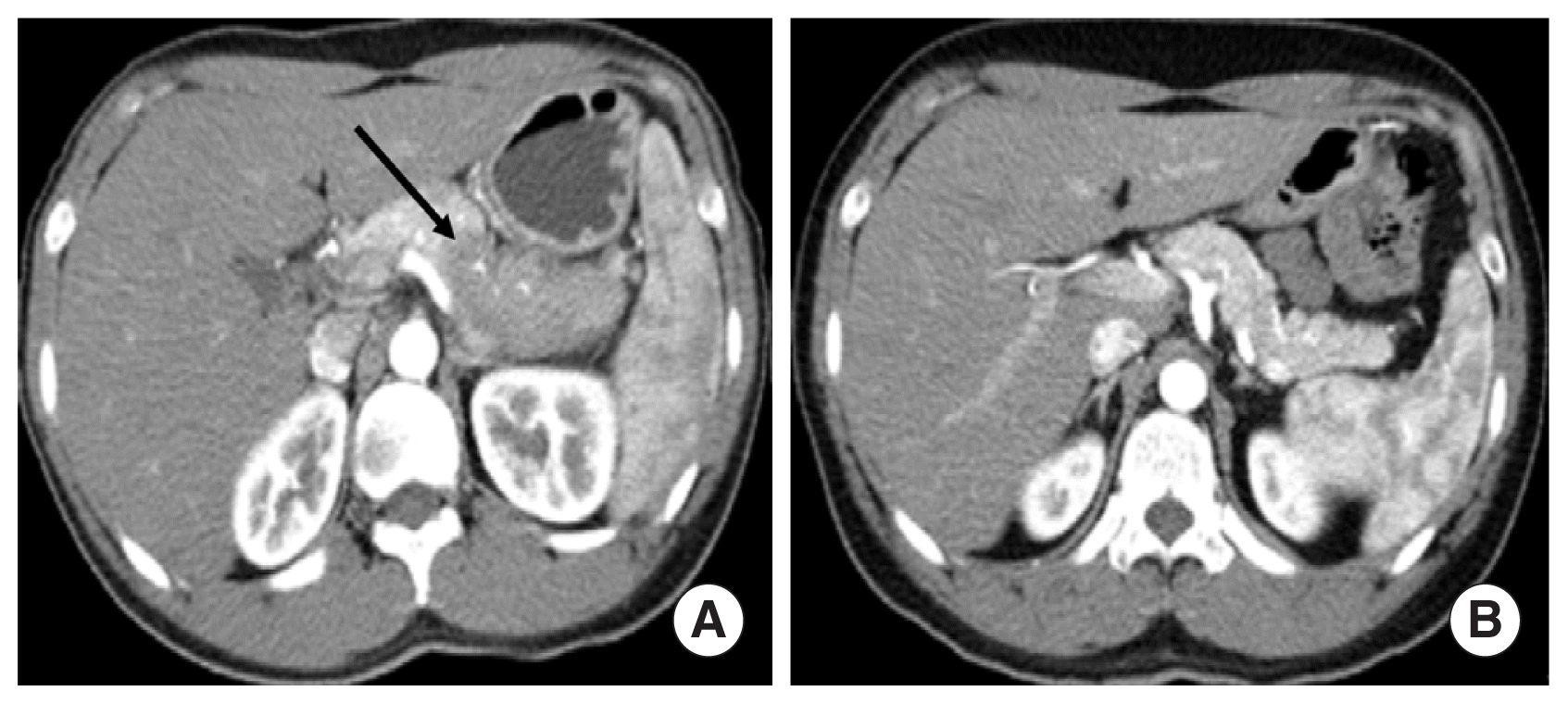
Staging workup including abdomen-liver 3 phase 3-dimensional CT, positron emission tomography (PET)-CT, esophagoduodenoscopy, and bone marrow biopsy was done. There was also involvement of lymphoma-like lesion in the pancreas on abdomen CT scan (Fig. 3A) and focal fluorodeoxyglucose (FDG) activity was showed on pancreas head and diffuse FDG uptake in tail on PET-CT but we did not perform a pancreas biopsy. There was no clear evidence of metastatic bone marrow involvement of lymphoma. Therefore, he was transferred to the oncology department and has been receiving chemotherapy including rituximab, cyclophosphamide, vincristine, and prednisone. His symptoms resolved and radiologic findings for pulmonary and pancreas lesions showed improvement after 3 cycles of chemotherapy (Fig. 3B).
DISCUSSION
We report a rare case of pulmonary MALT lymphoma presenting with diffuse bilateral bronchovascular thickening and focal GGO with concurrent pancreas involvement which is very uncommon. This patient showed successful response of treatment with cytotoxic chemotherapy and rituximab.
MALT lymphoma is a monoclonal lymphoid proliferation arising from a B-cell progenitor and is classified as low-grade marginal zone B-cell lymphoma of MALT in the World Health Organization classification [3].
Patients tend to present around the 6th decade, although rare cases in younger patients have been reported [4] (e.g., our patient was 43 years old). Development of some pulmonary MALT lymphomas has been reported to be associated with chronic inflammation of the bronchus associated lymphoid tissue and has been associated with multiple autoimmune diseases such as Sjögren syndrome, rheumatoid arthritis, common variable immunodeficiency, chronic inflammation from smoking, and infections (e.g., HIV and hepatitis C) [5]. Most patients who develop MALT lymphoma are former or active smokers [4]. While clinical manifestations are usually non specific and include cough, mild dyspnea, chest pain and occasionally hemoptysis, the majority of patients are asymptomatic, as in the presently reported case. One recent study evaluated 63 patients with pulmonary MALT lymphoma [6], with the median age being 60 years. Thirty-six percent of the patients had no symptoms at diagnosis, and 46% of them had at least one extrapulmonary location of lymphoma, which is most commonly found in the stomach area. However, there have been no reports of metastasis of the pancreas, as in our case.
Pulmonary MALT lymphoma is not easily diagnosed if patients show atypical or nonspecific radiological findings and clinical manifestations. Most frequent radiologic findings of pulmonary MALT lymphoma are nodules, masses, and/or consolidation, and the disease can be bilateral or unilateral [4]. Some patients present with nodular/mass-like areas of GGO or consolidation (or a mix of both), which tend to be in a bronchovascular distribution and often show on air bronchograms. More rarely, pulmonary MALT lymphoma manifests as patchy large areas of GGO throughout the lung [4]. Hilar and mediastinal lymphadenopathy can be present up to 30% of the time, while pleural effusions are uncommon (10%) [1]. Diffuse interstitial lung disease pattern presenting GGO is not a frequent finding and its incidence is 6%–10% in pulmonary MALT lymphoma [7]. Our patient showed diffuse bronchovascular bundle thickening in bilateral lung with very focal and subtle GGO with interlobular septal thickening. This finding is very rare and not easily differentiated from other interstitial pulmonary disease [2]. Differential diagnosis of pulmonary MALT lymphoma includes diseases with peribronchovascular distribution on CT such as sarcoidosis, Wegener’s granulomatosis, perilymphatic spread of metastatic malignancy, and infection [4].
Definitive diagnosis of pulmonary MALT lymphoma requires histological examination of biopsy specimens obtained via minimally invasive procedures, including transbronchial biopsy or percutaneous transthoracic core needle biopsy. However, the tissue specimen is occasionally insufficient for diagnosis, particularly in patients with atypical CT findings; therefore, surgical biopsies are sometimes needed. In addition, immunohistochemical staining for confirming a monoclonal B-cell population is crucial for accurate diagnosis [4].
Pulmonary MALT lymphoma has an indolent nature with the potential for spontaneous regression. There are two cases of spontaneous regression without any treatment [8,9]. Optimal treatment is currently with the chemotherapeutic agent chlorambucil for cases with disseminated disease [6]. The therapeutic role of immunotherapy using anti-CD20 monoclonal antibody (rituximab) is challenging, especially in MALT lymphoma cases expressing CD20 antigen. Rituximab is used either in combination with chemotherapy or as a single agent [10]. Our case also demonstrated immunoreactivity to CD20 on immunohistochemical staining and favorable response to chemotherapy along with rituximab, cyclophosphamide, vincristine, and prednisone. Surgery and radiation therapy are options available for localized disease and could be considered after staging and evaluation of all other mucosal sites have been performed [4]. The estimated 5- and 10-year overall survival rates were 90% and 72%, respectively [6].
In conclusion, pulmonary MALT lymphoma is a rare disease and cannot be confirmed easily in cases with atypical radiological findings. Definitive diagnosis begins with suspicion of pulmonary lymphoma followed by adequate tissue biopsy. We presented the case of a 43-year-old patient with surgical biopsy-proven diagnosis of pulmonary MALT lymphoma that had very atypical radiological findings. The patient was successfully treated with rituximab combined with cytotoxic chemotherapy.
ACKNOWLEDGMENTS
This work was supported by a Soonchunhyang University Research Fund.