Successful Complex Endovascular Intervention for a Patient with Typical Buerger’s Disease
Article information
Abstract
In thromboangiitis obliterans (Buerger’s disease), little progress has been made in its treatment. Medical treatment is ineffective and bypass surgery is possible only in limited case. Nowadays, endovascular intervention is popular treatment option for Buerger’s disease. Endovascular procedure is safe, technically feasible, and effective. Especially, in long occlusion lesion with distal channel, endovascular intervention can be an effective treatment option. Herein, we report a case of complete wound healing following the successful endovascular intervention in Buerger’s disease patient with distal channel.
INTRODUCTION
Buerger’s disease is chronic progression disease. Limb loss rate is significantly higher than atherosclerosis or other necrotizing immune arteritis [1]. Until now, the only proven treatment of preventing disease progression and avoiding amputation is smoking cessation [2]. Medical treatment is ineffective [3,4]. Bypass surgery is possible only in limited case because of diffuse distal involvement with no distal runoff [5]. But it has been controversies in the role of endovascular intervention. Herein, we report a case of complete wound healing following the successful endovascular intervention in Buerger’s disease patient with distal channel.
CASE REPORT
A 47-year-old male with 40 pack-year was presented for unhealed foot ulceration. Also, he could not lie asleep due to foot pain. When he was 35 years old, he was diagnosed with Buerger’s disease. He recommended to stop smoking repeatedly, but he kept smoking. Since then, he had been admitted repeatedly due to aggravated foot pain and wound. Finally, amputation for 4th and 5th left toes was performed. His compliance was very bad. It was passed for appropriate time for intervention. However, after amputation, foot pain lasted and necrotic wound was spread to remnant toes and dorsum.
On admission, pulses of the dorsalis pedis and posterior tibial artery were very weak. The ankle-brachial index of left foot was 0.23. Initial angiography showed the chronic total occlusion (CTO) from mid superficial femoral artery to distal anterior tibial artery portion. Also, left posterior tibial, peroneal, and dorsalis pedis artery were also totally occluded (Fig. 1). Angiography showed the chronic total occlusion from right superficial femoral artery to right dorsalis pedis. So the ankle-brachial index of right foot was non-measurable. But wound of right foot was insignificant.
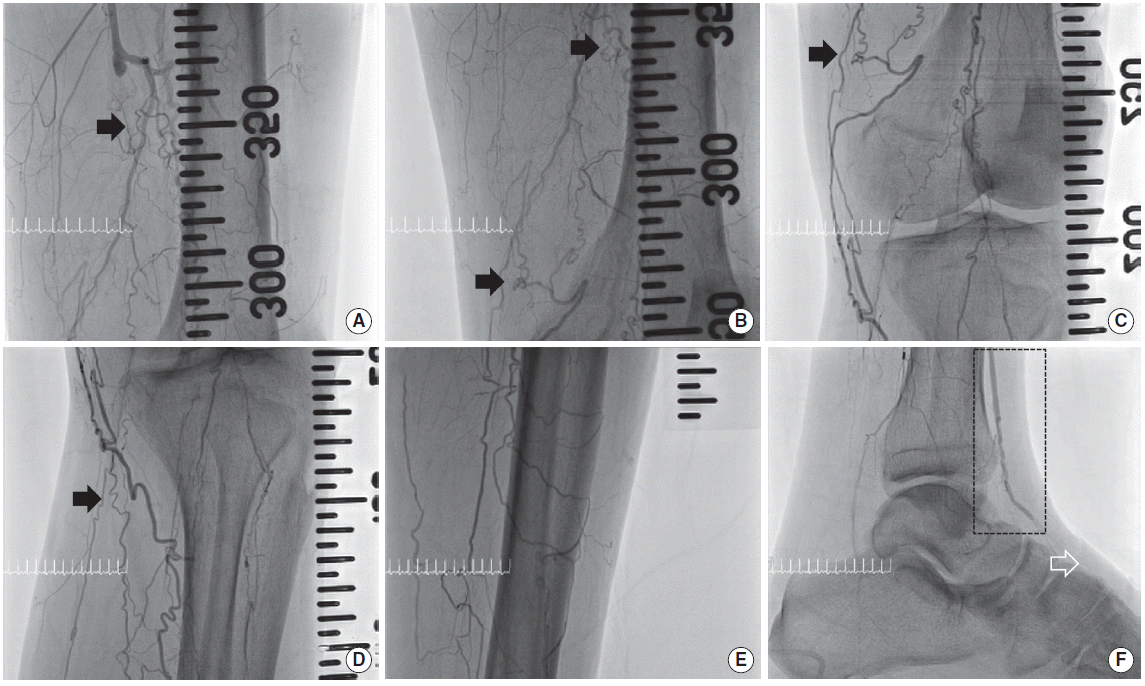
(A–F) Before endovascular intervention, initial angiography shows chronic total occlusion from mid superficial femoral artery to distal anterior tibial artery. Arrow: corkscrew appearance. Black box: distal channel of anterior tibial artery. White arrow: total occlusion of dorsalis pedis artery.
Bypass surgery could not become the treatment of choice since there was no distal runoff vessel available. So endovascular intervention was planned. Because patient could not keep supine position due to foot pain, the endovascular intervention was done under general anesthesia.
The length of target was very long. Also the target lesion was CTO and the bifurcation portion of the below the knee was totally occluded. However, he had distal channel on anterior tibial artery. It means he had patent distal part of anterior tibial artery. Because of the distal channel, both anterograde and retrograde approach was possible. After bi-lateral puncture at both left femoral artery and distal anterior tibial artery, 6Fr short sheath and CXI (COOK, Bloomington, IL, USA) microcatheter were inserted, respectively. Subintimal wire tracking using 014 wire such as Command-ES (Abbott, Abbott Park, IL, USA) supported by CXI was tried and was successfully passed into target lesion. After guidewire externalization, sequential ballooning with Sleek (Cordis, Fremont, CA, USA) 3.0×220 mm and Mustang (Boston Scientific, Marlborough, MA, USA) 4.0×40 mm was done up to maximal pressure at the portion of anterior tibial, popliteal, and superficial femoral lesion. And then, after successful guidewire passage into dorsal CTO lesion, sequential ballooning with Sleek 1.5×100 mm and Sleek 2.0×120 mm up to maximal pressure was done. After ballooning, balloon response was excellent without significant remnant stenosis except the entry portion of superficial femoral artery where there was remnant stenosis of more than 50%. So, in that site, stent deployment with Smart (Cordis) 6.0×40 mm was done without immediate complication. Adjuvant ballooning with Mustang 5.0×40 mm was done up to maximal pressure and residual stenosis was less than 20% (Fig. 2). Next, we planned to perform endovascular intervention for posterior tibial lesion. However, at this time, the blood pressure was dropped suddenly into 70/40 mmHg, of which the reason still remained unknown. Anyway, the staged endovascular intervention for the remnant lesion was planned and 1st procedure was stopped. After 7 days from 1st procedure, 2nd procedure for the CTO lesion of posterior tibial artery was tried. After 1st procedure, pain was subside dramatically. So 2nd procedure was done without general anesthesia. After 1st procedure, pain has been subsided dramatically. Therefore, after 7 days from 1st procedure, 2nd procedure for the CTO lesion of posterior tibial artery was done without general anesthesia.
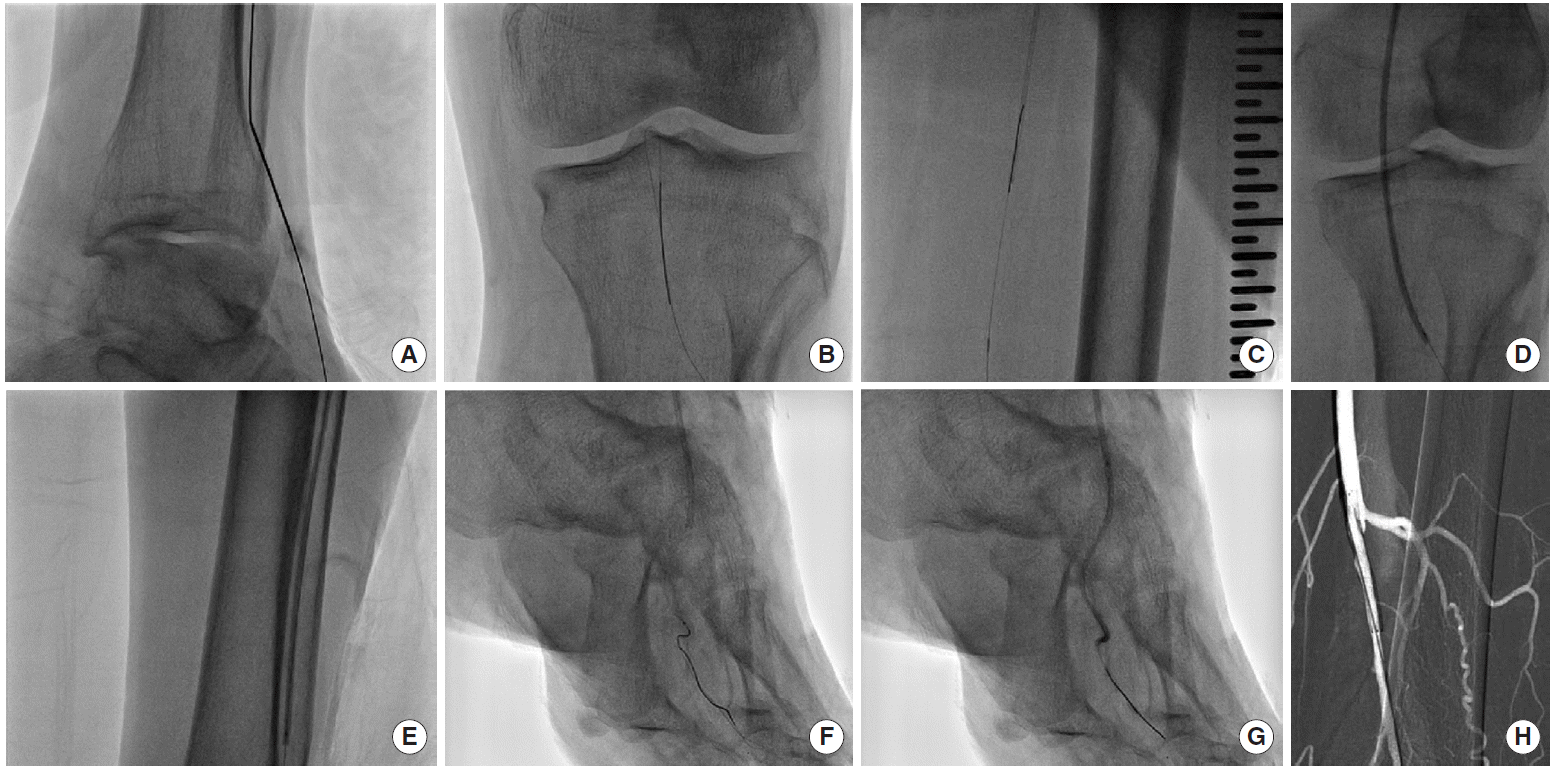
The picture shows the procedure. (A) Retrograde puncture through distal channel of anterior tibial artery. (B, C) Wire tracking. (D, E) Ballooning. (F, G) Dorsalis pedis artery wire tracking and ballooning. (H) Stenting.
He took aspirin 100 mg, cilostazole 200 mg, clopidogrel 75 mg, and rosuvastatin 10 mg. Only wound care such as dressing and debridement were served without skin graft. Wound was completely healed following about 7 months from complete procedure (Fig. 4).
DISCUSSION
Buerger’s disease is an inflammatory occlusive disorder affecting small and medium-size arteries and veins of young, male, smokers. The prognosis for patients with respect to limb loss is very bad [1]. Median prevalence of amputation, based on recently reported series, has been reported to be as high as 24.4% for minor amputations and 8.6% for major amputations, for a total amputation rate of 33% for patients treated conservatively [6].
At present, smoking cessation is the only strategy to prevent the progression of the disease and avoid amputation. Ohta et al. [7] reported that all patients who lost their limbs did not abstain from smoking. Unfortunately, the percentage of Buerger’s disease patients who maintain smoking cessation is very low. In one study, the continuous abstinence rate decreased from 29% at the end of treatment to 18.5% at 12-month follow-up [2].
Intravenous administration of a prostacyclin was more effective than oral aspirin in a randomized trial. Healing rate of ischemic lesions and pain relief were significantly higher in the prostacyclin group [8]. In contradiction, oral prostacylin did not confer any benefits in a placebo-controlled trial [9]. And there is no proven evidence for antiplatelet agents, anticoagulants, and calcium channel blockers in Buerger’s disease [3,4].
Bypass surgery for Buerger’s disease is possible only in a few case. It is related to the lack of potential distal targets for bypass. The disease is distal, diffuse, and segmental. In addition, because superficial venous structures are frequently involved in Buerger’s disease, the great and short saphenous veins unsuitable as a conduit for bypass. Therefore bypass surgery is technically challenging, with very low feasibility and patency rate [5].
Nowadays, endovascular intervention is popular treatment option for Buerger’s disease. Graziani et al. [10] reported that an endovascular procedure is safe, technically feasible, and effective. The report showed comparison between surgical revascularization and angioplasty. Angioplasty achieved high rates of technical success (94.7% vs. 10%), the decreased rates of amputation (16.6% vs. 19%), and the maintenance of clinical improvement during midterm follow-up periods (83.3% vs. 59%) [10].
In our case, although the lesion length was very long, the presence of the available distal channel enabled the guidewire to pass the target lesion by using the bi-directional approach with anterograde and retrograde puncture. After successful endovascular intervention, pain was subsided and wound was healed completely. In conclusion, the endovascular intervention can be an effective treatment option for the Buerger’s disease with optimal anatomy such as our case.

