INTRODUCTION
The myocardial injury occurs in a wide range of areas, and mediators such as nerve growth factor and neurotrophin-3 are overexpressed to increase cardiac sympathetic activity, which can lead to a ventricular electrical storm (VES) [1].
Although VES and other refractory ventricular arrhythmias (VAs) can be successfully treated with catheter ablation, it may be difficult to implement immediately in hemodynamically unstable patients [2]. The sympathetic nervous system is often responsible for ventricular tachycardia (VT) and is an effective therapeutic target. The autonomic tone can be controlled by surgical sympathetic denervation. Surgical sympathectomy, like catheter ablation, may be difficult to implement immediately in hemodynamically unstable patients.
The stellate ganglion block (SGB) is a minimally invasive technique performed to control VES through temporary sympathetic cessation [2]. Although it is not the primary treatment option, many reports have shown that SGB is effective in controlling refractory VAs.
Herein, we report the case of a patient with refractory VA, which was managed by the left and right SGB. The SGB was performed to control VES, after which the patient went through a successful laparoscopic nephrectomy. The patient provided written informed consent for the publication of clinical details and images.
CASE REPORT
A 59-year-old man with a past medical history of hyperlipidemia and hypertension came to the emergency room for chest pain. Electrocardiography revealed a non-ST elevation myocardial infarction. Subsequent coronary angiography with ballooning was performed, following which, a coronary artery bypass graft was planned.
The patient complained of dyspnea and his oxygen saturation dropped; he was intubated and shifted to the intensive care unit (ICU). On the second day of hospitalization, the patient had persistent VT and his consciousness decreased. Cardiopulmonary resuscitation and defibrillation were performed, and a return of spontaneous circulation was achieved.
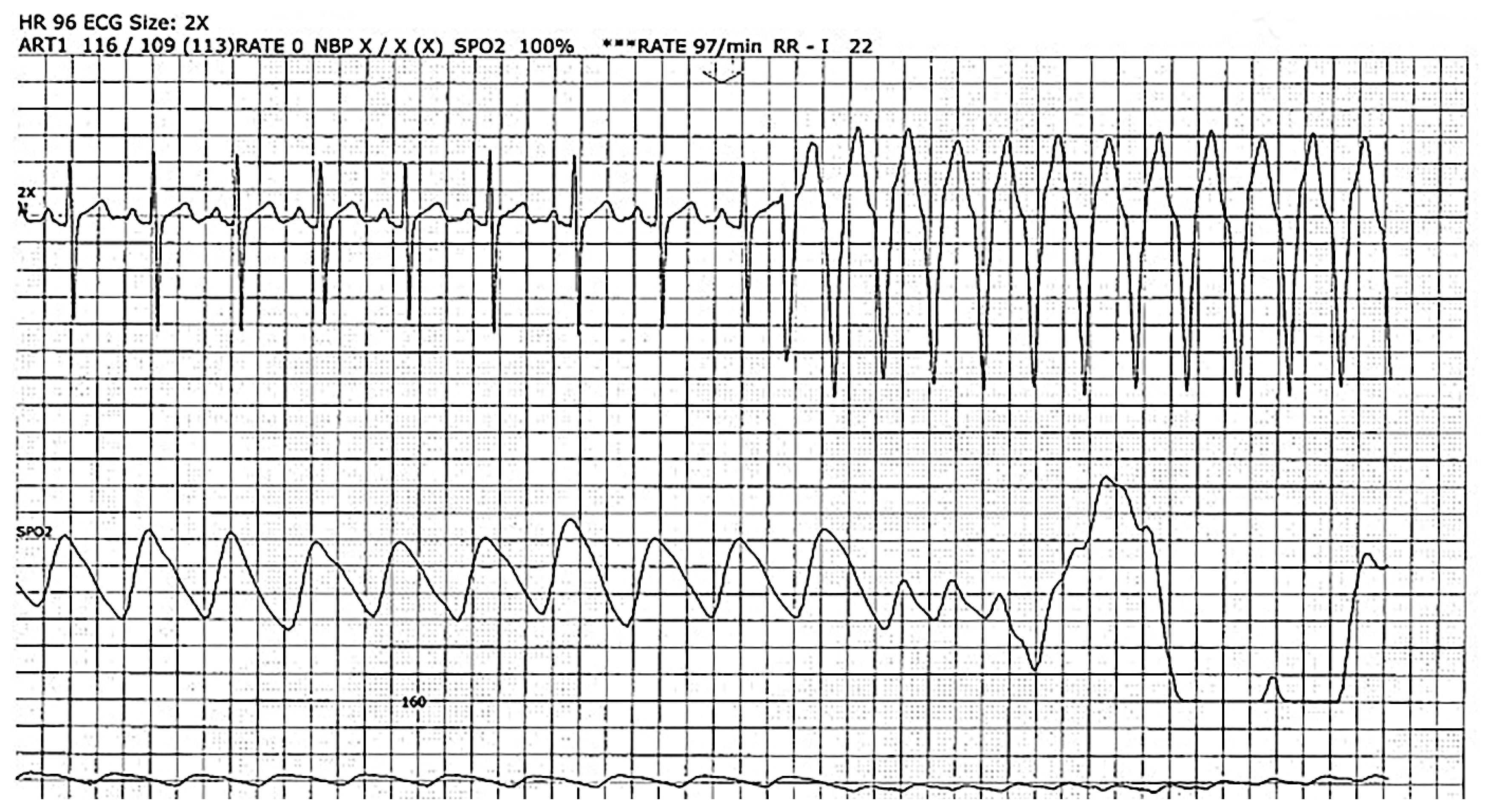
As a result of frequent VT, the implantation of a temporary pacemaker and implantable cardioverter-defibrillator (ICD) was done. Drugs such as antiarrhythmics, anticoagulant, isosorbide dinitrate, and amiodarone were administered. In addition, anti-tachycardia pacing with ICD cardioversion was performed; however, VT persisted (Fig. 1). Subsequently, our pain clinic was consulted for a temporary sympathetic blockade. The SGB was performed three times for 3 days in a row.
The patient was placed in a supine position and his neck was supported by placing a small pillow under his shoulder. Initially, the SGB was performed on the left side; however, both unilateral and bilateral were considered according to the patient’s response. The C6 anterior tubercle was palpated with the left hand, and 1% mepivacaine 6 mL was injected. The vital signs stabilized after the procedure, and no other neurological abnormalities were observed. After the first SGB, VT episodes—which occurred 3–4 times in 24 hours—decreased to approximately one.
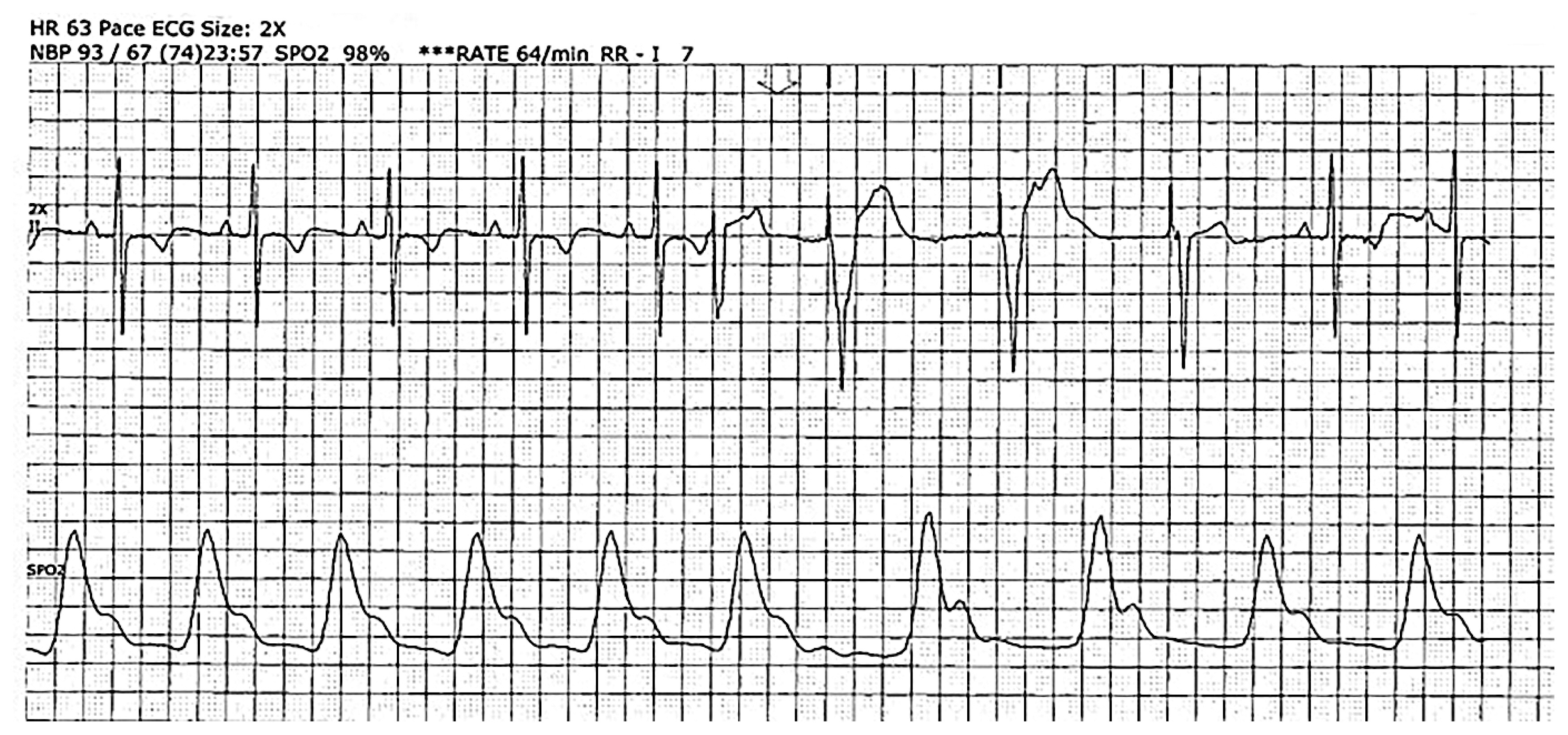
On the next day, an ultrasound-guided second SGB was performed on the right side. The patient was then placed again in the supine position. After confirming the C6 transverse process with the help of ultrasound guidance, the transducer was slightly moved caudally for the needle to insert toward the longus colli muscle, above the C7 transverse process. The needle was slowly inserted while avoiding the C6–7 nerve and internal jugular vein, subsequently 1% mepivacaine 6 mL was injected. There were no side effects other than a slightly hoarse voice; the vital signs were stable. VT episodes did not occur for 24 hours after the second SGB; the sedative drugs were slowly tapered off as the patient’s condition stabilized. The next day, a second SGB was performed on the right side and finally on the left side in the same way. After a week, VT episodes resolved, and the patient was transferred from the ICU to the general ward (Fig. 2).
During hospitalization, an incidental left renal mass was identified on the chest computed tomography. Subsequently, a laparoscopic radical nephrectomy was planned. To prevent stress-induced VT, left SGB was administered before general anesthesia, and the surgery was performed with the ICD stopped. There were no VAs, and the patient’s vital signs remained stable. The surgery was successful, and the patient recovered uneventfully.
DISCUSSION
VES is triggered by three factors: cardiomyopathy, ischemia-like triggers, and autonomic dysregulation (AD) such as decreased parasympathetic input and sympathetic hyperinnervation. AD occurs due to sprouting and perturbed innervation of sympathetic nerves, generating VT, and increasing beta–adrenergic receptor density. Therefore, autonomic modulation is an important factor in the treatment of VESs. Cardiac sympathetic ganglia include cervical, stellate, and thoracic ganglia. Moreover, it includes innervation from the atria to the ventricle and from the base to the apex of the heart and can be a target for autonomic modulation [1].
Autonomic modulation includes a thoracic epidural block (TEB), SGB, and open cardiac sympathetic denervation [2].
The TEB can block the T1–T4 level among cardiac sympathetic innervations, including C1 to T4. Previous studies reported that VES could be effectively prevented when bupivacaine was injected at 2 mL/hr, by placing a catheter in the thoracic epidural space [3].
However, it is inefficient to induce a block of sympathetic nerves presenting in the front and side of the spinal body, by administering drugs into the posterior epidural space. While maintaining the catheter, side effects such as bleeding (related to the use of anticoagulants and antiplatelet drugs) and infection may occur. Although thoracic sympathetic nerve block can be performed directly, its access is difficult due to the anatomical location, and side effects such as pneumothorax may occur, which makes the procedure difficult.
The surgical approach of left cervical sympathetic denervation is also possible and is known to be effective against idiopathic or polymorphic VT [2]. However, it is not commonly performed because of the difficulty of the procedure and its side effects.
SGB is effective when bilaterally performed, and both blind and ultrasound-guided techniques can be used [4,5]. Using ultrasound guidance, local anesthetics can be injected into the anterior tubercle of the transverse process of the C6 vertebra or the anterior surface of the longus colli muscle of the C7 vertebra with minimal complications such as accidental intravascular injection and nerve damage [6].
Furthermore, the bilateral SGB procedure acutely reduces monomorphic and polymorphic VT and is recommended to be performed under ultrasound guidance [7]. When SGB is performed for VES treatment, the VES burden can be seen to decrease after 2 doses, and cases of complete remission after 3–4 or so trials have been reported.
In 2016, the clinical protocol for VES was introduced by Duke’s neurocardiology service (Fig. 3).
The present case was treated with SGB for VES that occurred after non-ST elevation myocardial infarction, and then prophylactically SGB before nephrectomy surgery to safely complete the operation.
For SGB, the treatment dose or the number of treatments according to the disease remains unclear. There are no guidelines for the number or dose of SGB for VES treatment yet, and further large-scale studies are needed for this. SGB is a safe and effective treatment option for VES.