SMS 2012 June;18(1):56-60.
Published online 2012 July 17 |
| Copyright ⓒ 2010 Soonchunhyang Medical Science
|
| Complete Remission of Refractory Hepatic Hydrothorax in Patient with Advanced Liver Cirrhosis and Hepatocellular Carcinoma Using Transjugular Intrahepatic Portosystemic Shunt |
| Hyeon Jeong Goong, Sang Gyune Kim, Seung Sik Park, Youn Hee Cho, Young Seok Kim, Boo Sung Kim |
| Institute for Digestive Research, Department of Internal Medicine, Soonchunhyang University College of Medicine, Bucheon, Korea |
| Corresponding Author: Sang Gyune Kim , Tel: +82-32-621-5071 , Fax: +82-32-621-5080 , Email: mcnulty@schmc.ac.kr
|

|
ABSTRACT
|

|
|
| Hepatic hydrothorax is defined as the presence of pleural fluid (>500 mL) in the absence of primary cardiac or pulmonary disease. Initial treatments consist of a low salt diet, diuretics, and thoracentesis. If these are not effective, other modalities should be considered. The transjugular intrahepatic portosystemic shunt (TIPS) placement is one of the modalities for treatment of hepatic hydrothorax. However, the effects of TIPS placement have been contradictory. A 42-year-old man was diagnosed hepatic hydrothorax with liver cirrhosis. He was managed with medical therapy, but it was not effective to control hepatic hydrothorax. This case is reported with a review of relevant literature. |
|
Keywords: Refractory hydrothorax; Transjugular intrahepatic portosystemic shunt |
|

|
INTRODUCTION
|

|
|
| Hepatic hydrothorax is defined as the presence of pleural fluid (>500 mL) in the absence of primary cardiac or pulmonary disease. It occurs in approximately 5% to 10% of patients with advanced cirrhosis. In most cases, it is associated with ascites. However, there have been some cases those are not related to ascites. The possible causes include hypoalbuminemia, hypertension of the azygous vein, defects in the tendinous portion of the diaphragm, and lymphatic leakage through the thoracic duct. It is mostly rightsided (85%), but may also be left sided (13%) or bilateral (2%). Initial treatments consist of a low salt diet, diuretics, and thoracentesis. If these are not effective, other modalities should be considered. For example, pleurodesis, surgical repair of the diaphragmatic leakage, insertion of a peritoneovenous shunt, and liver transplantation has been tried. The transjugular intrahepatic portosystemic shunt (TIPS) placement is one of the modalities for treatment of hepatic hydrothorax. However, the effect of TIPS was contradictory. We report here a case of hepatic hydrothorax that was treated with TIPS. |

|
CASE REPORT
|

|
|
A 42-year-old man visited our hospital for moderate dyspnea. The patient has been diagnosed with liver cirrhosis in combination with chronic hepatitis B for 10 years. Hepatocellular carcinoma (HCC) was found on regular follow-up 3 years previously, for which he received transarterial chemoembolization (TACE), and then radiofrequency ablation (RFA). The recurrence of HCC did not happen after the treatment. At that time, he already had ascites and esophageal varix, and had complained of intermittent dyspnea because of right hydrothorax for 1 year. Esophageal varix was treated with three times of endoscopic variceal band ligation and the last esophagogastroduodenoscopy showed F1 and Cw of esophageal varix. He had had no previous hepatic encephalopathy. This symptom was repeated despite medical treatment and could be relieved only by repeated thoracentesis.
At admission, the patient’s blood pressure was 120/80 mmHg, pulse rate was 78 beats/min, respiratory rate was 25 breaths/min, and body temperature was 36°C. Physical examination identified a reduced lung sound and percussion dullness in right lung field.
Peripheral blood test results indicated that the patient’s white blood cell count was 2,900/μL, hemoglobin level was 8.1 g/dL, platelet count was 50,000/μL, and prothrombin time (PT) was 19.5 seconds (PT ratio [PTR], 42%; international normalized ratio [INR], 1.74). Blood chemistry tests as followed: aspartate aminotransferase, 60 IU/L; alanine aminotransferase (ALT), 40 IU/L; alkaline phosphatase, 186 IU/L; total bilirubin, 2.48 mg/dL; total protein, 6.8 g/dL; albumin, 2.4 g/dL; creatinine, 1.0 mg/dL; and lactate dehydrogenase (LDH), 430 IU/L. The Child-Pugh score was 10 points and the Model for End-stage Liver Disease (MELD) score was 16 points. In pleural fluid analysis, the LDH level was 79 IU/L, protein was 835 mg/dL (8.35 g/dL), and albumin was 0.4 g/dL. Pleural fluid/serum total protein ratio was greater than 0.5, pleural fluid/serum LDH ratio was less than 0.6, and pleural fluid LDH was less than 200 U/L. According to Light’s criteria, exudate was evident, but serum-pleural fluid albumin gradient was 2.0 g/dL. So we decided it was transudate. A pleural acid-fast bacillus smear and bacterial culture were negative, and no malignant cell was detected. A simple chest X-ray and computed tomography showed significant right-sided hydrothorax
(Fig.1)
. Tumor was shown as post RFA necrosis on S4 and S6. There was no portal vein thrombosis (Fig. 2A). TIPS placement was performed at once (Fig. 2B). After the procedure, the value of wedge hepatic venous pressure (WHVP) was decreased from 17 to 10 mmHg. The amount of pleural fluid decreased remarkably, as determined by a simple chest X-ray. The patient’s symptoms also improved, and he was discharged with a drug regimen of furosemide (80 mg) and amiloride (20 mg). During follow-up, there was no sign of TIPS associated hemolysis.
One year after TIPS placement, the patient again visited the hospital due to relapsed dyspnea. A blood test found a total bilirubin level of 4.3 mg/dL and PT of 23.6 seconds (PTR, 35%; INR, 2.12). Hepatic venography showed 75% stent thrombosis. The patient underwent angioplasty for TIPS revision. Thereafter, the WHVP was reduced from 16 to 5 mmHg and the patient’s symptoms improved. Also the hydrothorax was well controlled
(Fig.3)
.
During follow-up period, the patient was admitted due to hepatic encephalopathy and received TACE due to a recurrence of hepatocellular carcinoma at 6 months after TIPS placement. He received 2nd balloon angioplasty for stent restenosis at 8 months after the first revision. He died of hepatic failure resulting from an aggravation of HCC. |

|
DISCUSSION
|

|
|
Hepatic hydrothorax is one of several complications arising from portal hypertension in liver cirrhosis. In most cases, it is related to ascites. Transudate is thus evident on thoracic fluid analysis and the serum-pleural fluid albumin gradient is >1.1 g/dL. If combined with bacterial infection, spontaneous bacterial empyema can occur
[1]
. Patients complain of dyspnea, chest pain, cough without sputum, fatigue from hypoxia, and so on. Thus, optimal treatments are required, but usually not effective as much as we expect. As in ascites, medical treatments such as a low salt diet and diuretics are commonly used. When it doesn’t work at maximal tolerated dose, this condition is defined as refractory hepatic hydrothorax, and surgical procedures should be considered. However, repetitive thoracentesis and chest tube drainage might worsen the patient’s quality of life and cause hyponatremia and hypoalbuminemia due to volume loss
[2]
. Pleurodesis and peritoneovenous shunting cause rapid fluid re-accumulation and procedure-related complications
[3]
. The only curative treatment for hepatic hydrothorax is liver transplantation.
TIPS is one of the treatments for reducing portal hypertension, which is an important cause of hepatic hydrothorax. It can decrease the porto-systemic pressure gradient and limit the retention of sodium and water. The treatment of hepatic hydrothorax with TIPS was first described by Strauss et al. in 1994
[4]
and first reported by Gordon et al. in 1997
[5]
. Several subsequent studies have reported the effects of TIPS placement
[6-8]
. Wilputte et al.
[9]
reported that TIPS placement can cause a variety of procedure-related complications, and did not improve the overall prognosis in some patients. The results of some studies are summarized in
Table 1
. Complications reported to date include: puncture and fistula, TIPS thrombosis (10% to 15% in the immediate post-procedural period), TIPS stenosis (70% within the first year), hepatic encephalopathy (20% to 30%), TIPS-induced hemolysis (uncovered stent, 10%), TIPS-associated infection (1.3%), cardiac failure (2.5%), liver failure (1.9%), and hepatorenal syndrome (4.3%)
[10]
. TIPS-related causes of death include: liver failure, acute respiratory distress syndrome, renal failure, and sepsis. Thus, the use of a TIPS is not appropriate in all cases. TIPS placement should not be considered in patients who have poor prognostic factors, such as advanced age (>60 years); emergent procedure; high ALT (>100 IU/L), bilirubin (>3 mg/dL), and/or creatinine level; or previous hepatic encephalopathy. Also, the clinical response is related to overall survival and mortality
[11]
. The 30-day mortality and 1-year survival rates in patients without poor prognostic factors who receive TIPS are good [11,12]. Recent studies have reported that pre-procedural MELD score is related to postprocedural outcome, overall survival and mortality rate. Previous studies have suggested that MELD score to predict poor prognosis is >18, and Child-Pugh score is >11
[13]
. However, recently the cut off value of the score tend to be getting low [11,14]. Also, MELD score is prefered to predict outcome
[15]
.
In this case, unfortunately we could not perform any tests to evaluate diaphragmatic defects. The patient had no poor prognostic factors (no previous hepatic encephalopathy, age 65 years and serum bilirubin level <3 mg/dL). Also, the MELD score was less than 18 and the Child-Pugh score was less than 11 points. So, he was not at high risk. After TIPS he showed complete clinical response. He survived for 20 months, and the amount of hydrothorax was controlled well. However, the patient developed some complications, such as elevated bilirubin level, hepatic encephalopathy, and stent thrombosis. He died due to the recurrence of hepatocellular carcinoma. |
|

|
FIGURES
|

|
|
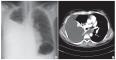
|
Fig.1
Initial chest X-ray (posterior-anterior) (A) and contrast-enhanced chest computed tomography (B) showing significant right pleural effusion. |

|

|
Fig.2
(A) Contrast-enhanced abdomen computed tomography showing post radiofrequency ablation necrosis on S4, S6 and no definite portal vein thrombosis. (B) Transjugular intrahepatic portosystemic shun placement was performed. |

|

|
Fig.3
Simple chest X-rays after 1 year showing well-controlled hydrothorax. |

|
|

|
TABLES
|

|
|

|
Table.1
Transjugular intrahepatic portosystemic shunt (TIPS) for the treatment of hepatic hydrothorax |

|
|
| |

|
REFERENCE
|

|
|
|
1.
|
Kiafar C, Gilani N. Hepatic hydrothorax: current concepts of pathophysiology and treatment options. Ann Hepatol 2008;7:313-20. |
|
2.
|
Dumont AE, Mulholland JH. Flow rate and composition of thoracic-duct lymph in patients with cirrhosis. N Engl J Med 1960;263:471-4. |
|
3.
|
Falchuk KR, Jacoby I, Colucci WS, Rybak ME. Tetracycline-induced pleural symphysis for recurrent hydrothorax complicating cirrhosis. A new approach to treatment. Gastroenterology 1977;72:319-21. |
|
4.
|
Strauss RM, Martin LG, Kaufman SL, Boyer TD. Transjugular intrahepatic portal systemic shunt for the management of symptomatic cirrhotic hydrothorax. Am J Gastroenterol 1994;89:1520-2. |
|
5.
|
Gordon FD, Anastopoulos HT, Crenshaw W, Gilchrist B, McEniff N, Falchuk KR, et al. The successful treatment of symptomatic, refractory hepatic hydrothorax with transjugular intrahepatic portosystemic shunt. Hepatology 1997;25:1366-9. |
|
6.
|
Degawa M, Hamasaki K, Yano K, Nakao K, Kato Y, Sakamoto I, et al. Refractory hepatic hydrothorax treated with transjugular intrahepatic portosystemic shunt. J Gastroenterol 1999;34:128-31. |
|
7.
|
Kidokoro H, Kanazawa H, Nachi T, Narahara Y, Osada Y, Mamiya Y, et al. A case of refractory hepatic hydrothorax successfully treated with transjugular intrahepatic portosystemic shunt. Nihon Shokakibyo Gakkai Zasshi 2003;100:707-12. |
|
8.
|
Haskal ZJ, Zuckerman J. Resolution of hepatic hydrothorax after transjugular intrahepatic portosystemic shunt (TIPS) placement. Chest 1994;106: 1293-5. |
|
9.
|
Wilputte JY, Goffette P, Zech F, Godoy-Gepert A, Geubel A. The outcome after transjugular intrahepatic portosystemic shunt (TIPS) for hepatic hydrothorax is closely related to liver dysfunction: a long-term study in 28 patients. Acta Gastroenterol Belg 2007;70:6-10. |
|
10.
|
Wong F. The use of TIPS in chronic liver disease. Ann Hepatol 2006;5:5-15. |
|
11.
|
Dhanasekaran R, West JK, Gonzales PC, Subramanian R, Parekh S, Spivey JR, et al. Transjugular intrahepatic portosystemic shunt for symptomatic refractory hepatic hydrothorax in patients with cirrhosis. Am J Gastroenterol 2010;105:635-41. |
|
12.
|
Rössle M, Gerbes AL. TIPS for the treatment of refractory ascites, hepatorenal syndrome and hepatic hydrothorax: a critical update. Gut 2010; 59:988-1000. |
|
13.
|
Ferral H, Gamboa P, Postoak DW, Albernaz VS, Young CR, Speeg KV, et al. Survival after elective transjugular intrahepatic portosystemic shunt creation: prediction with model for end-stage liver disease score. Radiology 2004;231:231-6. |
|
14.
|
Owen AR, Stanley AJ, Vijayananthan A, Moss JG. The transjugular intrahepatic portosystemic shunt (TIPS). Clin Radiol 2009;64:664-74. |
|
15.
|
Angermayr B, Cejna M, Karnel F, Gschwantler M, Koenig F, Pidlich J, et al. Child-Pugh versus MELD score in predicting survival in patients undergoing transjugular intrahepatic portosystemic shunt. Gut 2003;52:879-85. |
|
16.
|
Jeffries MA, Kazanjian S, Wilson M, Punch J, Fontana RJ. Transjugular intrahepatic portosystemic shunts and liver transplantation in patients with refractory hepatic hydrothorax. Liver Transpl Surg 1998;4:416-23. |
|
17.
|
Siegerstetter V, Deibert P, Ochs A, Olschewski M, Blum HE, Rössle M. Treatment of refractory hepatic hydrothorax with transjugular intrahepatic portosystemic shunt: long-term results in 40 patients. Eur J Gastroenterol Hepatol 2001;13:529-34. |
|
18.
|
Spencer EB, Cohen DT, Darcy MD. Safety and efficacy of transjugular intrahepatic portosystemic shunt creation for the treatment of hepatic hydrothorax. J Vasc Interv Radiol 2002;13:385-90. |
|
|
|