INTRODUCTION
Impetigo herpetiformis (IH) is a rare pustular dermatosis that typically occurs in pregnant women and rapid resolution in the postpartum period [1–3]. It is clinically and histologically consistent with pustular psoriasis. This similarity has led authors to name the disease ‘pustular psoriasis of pregnancy [2].’ According to the literature, although the causes of IH are still unknown, maternal complications like hypoparathyroidism, hypocalcemia, tetany, and delirium are reported in patients [4–7]. Generally the patients recovers after delivery, but IH frequently recurs in subsequent pregnancy with increased severity [8–10]. Maternal deaths are rare but there are risks of small for gestational age and stillbirth [3]. Early diagnosis and proper treatment is important in order to get a better prognosis for both the mother and baby.
CASE REPORT
A 33-year-old Korean two multigravida was admitted to Soonchunhyang University Cheonan Hospital because of pustular lesions in the third trimester. She had no history of hypertension, diabetes mellitus, and any other medical illnesses. She was non-smoker, and had not taken any drugs including indomethacin, oral pill, lithium, beta-blockers, which could be risk factors for pustular psoriasis. She had history of dermatosis especially psoriasis but no history of other skin diseases. The lesions of psoriasis occurred 10 years old for the first time. The size of the lesions occurred mainly in the upper body was about 1 cm. Lesions was worse when general condition was poor and applying the steroids immediately healed. The disease had not been worsen when the first pregnancy two years ago. But the disease had deteriorated second pregnancy at nearly 29 weeks’ gestation. Gradually, the skin lesions covered her entire body except the scalp, face, oral mucosa and palmoplantar regions. The symptom was pruritus, pustules, redness, and mild pain. But there are no fever, nausea, tachycardia, seizure, and other symptoms by then. Infectious disease medicine and dermatology were consulted. We treated her with topical steroids and emollients. Fetal surveillance with regular ultrasonogram and weekly nonstress tests showed a reassuring fetal status. But she felt fever, nausea, and severe pain in both thighs at 34 weeks’ gestation. Fetal estimated weight was 1,800 g, which is compatible with 31 weeks’ gestation. The patient also had oligohydramnios (amniotic fluid index=5 cm) on transabdominal ultrasound and minimal variability by nonstress test. We doubted small for gestational age caused by uteroplacental insufficiency. So we decided to deliver the fetus because there is a risk of stillbirth.
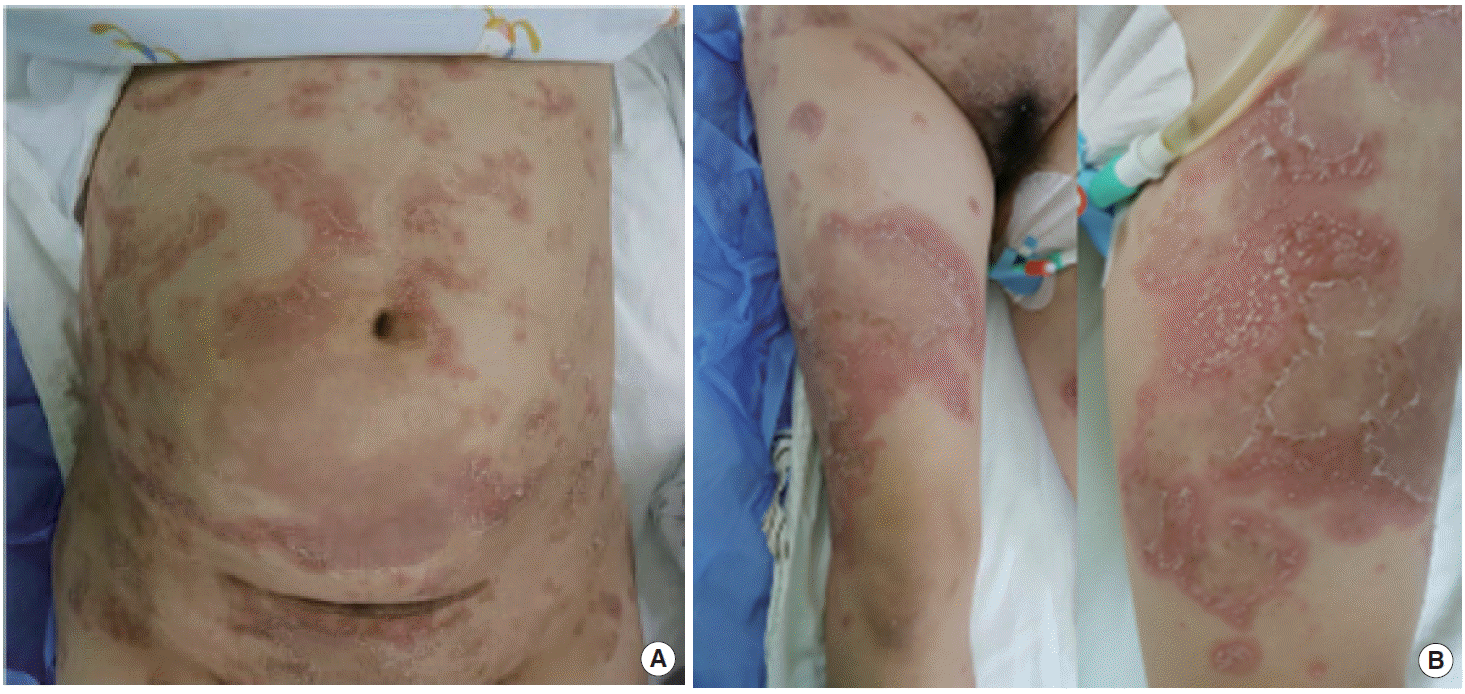
Her skin condition showed erythematous, scaly, and figurative lesions with pustules (Fig. 1). On physical examination, the patient had a body weight of 74 kg, a blood pressure of 110/70 mmHg, a pulse rate of 110 beats/min, a respiratory rate of 20 breaths/min, and a body temperature of 37.4°C. On serum biochemistry, the patient showed sodium 137 mEq/L (range, 136 to 145 mEq/L), chloride 100 mEq/L (range, 96 to 110 mEq/L), potassium 3.6 mEq/L (range, 3.5 to 5.1 mEq/L), and bicarbonate 27.1 mEq/L (range, 21 to 31 mEq/L). Moreover, the patient showed hemoglobin was 10.3 g/dL (range, 12.0 to 16.0 g/dL), white blood cell 14.5 10^3/μL (range, 4.0 to 10.8 10^3/μL), platelet 261/mm3 (range, 130 to 400/mm3), elevated C-reactive protein (CRP) 35.35 mg/L (range, 0 to 2.5 mg/L), decreased calcium 6.0 mg/dL (range, 8.0 to 10.5 mg/dL), and 25-OH vitamin D 8.0 ng/mL (range, 9.5 to 55.5 ng/mL). Coagulation profiles, parathyroid hormone, and serum albumin were within normal limits. The patient was negative for hepatitis B, syphilis, and human immunodeficiency viruses.
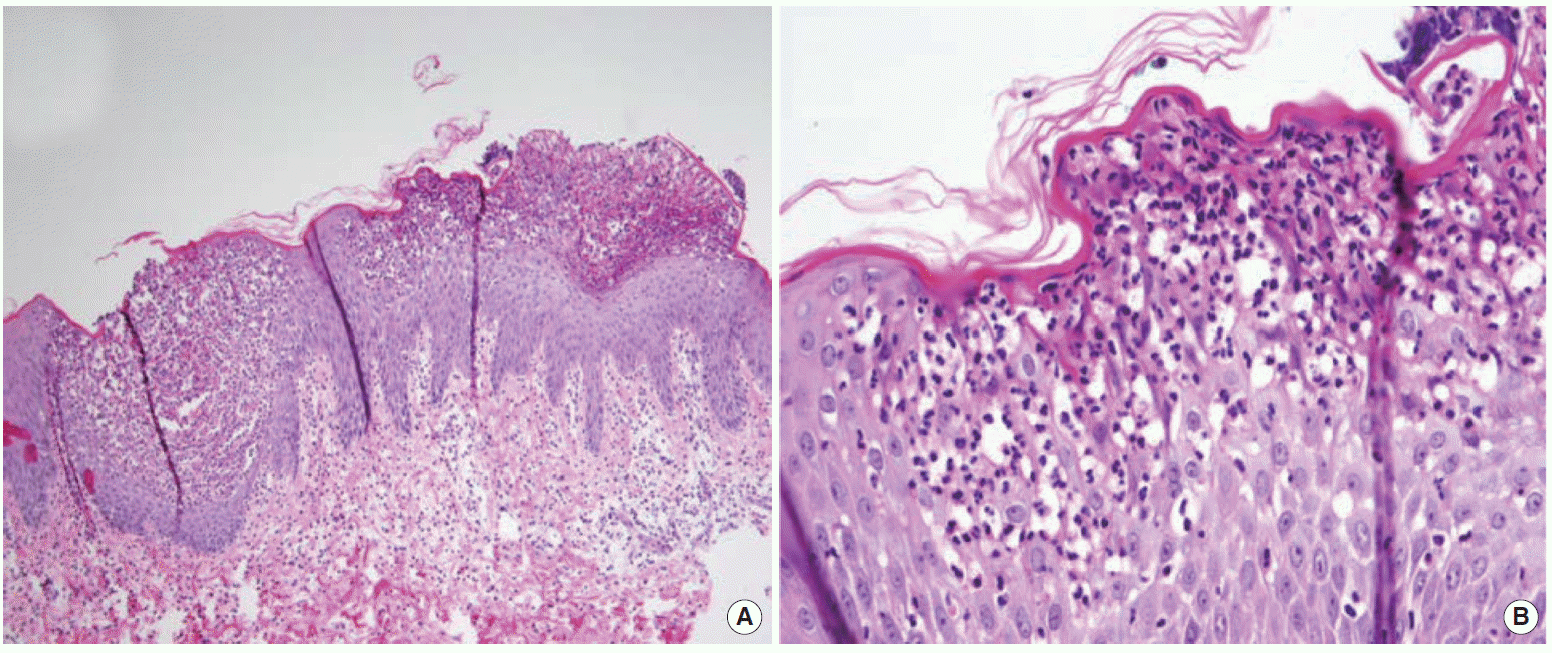
A healthy 1,790 g, Apgar score of 9 at five minutes, female baby with no pustule or erythema was born by cesarean section a day after admission. General anesthesia had been administrated to her who had no airway edema. The result of her punch skin biopsy showed compatible to IH (Fig. 2). The result of placenta pathology was not significant. The baby was sent to the neonate intensive care unit because of preterm birth and small for gestational age. After delivery, intravenous methylprednisolone (dose of 125 mg per day) was started immediately and used throughout hospitalization. After then intravenous methylprednisolone was changed to oral prednisone (dose of 15 mg per day). After expectant treatment for 4 days, skin lesions improved progressively and pain disappeared slowly. Ten days after delivery, her skin lesions on her trunk and arms had worsen rapidly. She presented with an active red multiple polycystic elevated lesion composed of peripheral micropustules and foul odor. So she was treated with a large dose of acitretin (dose of 60 mg per day) and cephalosporin antibiotics because she refused therapy using for cyclosporine or methotrexate. Three months after her delivery, her skin was returned to original state, except for residual pigmentation. Her baby was healthy, too.
DISCUSSION
Psoriasis is a chronic skin condition with a universal prevalence of 1% to 3% [4,5]. IH was first described by von Hebra in 1872, with a report of five pregnant women with erythematous plaques bordered by micropustules [3]. The etiology and pathogenesis of IH is still unknown. High level of progesterone during last trimester of pregnancy may be the triggering factor in the onset of the disease. Clinical examination is characterized by lesions commonly initiated on the thighs and groin, with centrifugal spread, in some cases affecting the mucous membranes [6,7]. The diagnosis of IH is mainly based on typical clinical findings in the third trimester of pregnancy. A disseminated spread of sterile pustules is often complicated by major general symptoms such as fever, nausea, and diarrhea [8]. Some cases report that IH may be complicated by gestational hypertension, diabetes, and fetal distress because of placental insufficiency [9]. Even though there was no sign of infection, laboratory findings indicate that leukocytosis, in particular, increased neutrophils and CRP. Cases of IH associated with hypocalcemia, hypoalbuminemia, and low serum levels of vitamin D have been reported [5–10], but the contribution of these abnormalities is still uncertain. This patient showed occurrence of third trimester of pregnancy and systemic symptoms like fever, nausea, and pain. And laboratory findings showed increased CRP, low serum level of vitamin D, and hypocalcemia. In this case, fetus showed small for gestational age and oligohydramnios. Some authors infer that IH may be an autoimmune disease. Although the significance of histopathological examination of skin and placenta is not specified clearly, it can help the clinician to diagnose IH [11–14]. Our result of pathology of skin was in concordance with IH.
There are no standard guidelines for treatments of IH. According to the literatures’ reviews, the treatments of IH mainly include systemic corticosteroids, cyclosporine, antibiotics, methotrexate, and systematic retinoids [15–19]. At the beginning of IH, corticosteroids are usually efficacious at a low dose of 20 mg a day. In some refractory cases, it is necessary to increase the its dose to 80 mg a day [16,19]. In progressive and unresponsive cases to corticosteroid therapy, oral cyclosporine is the second-line therapy for IH and antibiotics should be administered to treat or prevent secondary infection. Furthermore, it is reported that taking parenteral calcium, vitamin D, infliximab, and pyridoxine in high doses, is effective for IH during pregnancy. Moreover, taking methotrexate or retinoid (such as isotretinoin and acitretin) is helpful for curing IH after delivery [16,18,20]. In this case, we used the acitretin because she did not respond to corticosteroid therapy and refused therapy using for cyclosporine or methotrexate. Differential diagnoses include autoimmune bullous disease, dermatitis herpetiformis, pustular psoriasis, erythema multiforme, and gestational pemphigoid [5,8]. Although lesions tend to disappear after delivery, there is a risk of recurrence in subsequent pregnancies, presenting earlier and with more severe intensity. The current use of steroid and antibiotic therapy has powerfully reduced maternal deaths. However, the risk of stillbirth and perinatal mortality remains high, due to placental insufficiency, premature rupture of membranes, preterm labor, and small for gestational age. Clinicians should be aware of the possibility of sudden deterioration of the lesions and intrauterine fetal death associated with IH. IH is associated with high mortality and morbidity in fetus, but with early diagnosis and proper treatment a better prognosis could be achieved. In spite of many therapies, the effective treatments of IH are still not specified. Therefore, we have to establish a standardized diagnosis and treatment protocol in the future.